Draft!
Sections
A. Ordinal Classification
B. The suborders of Hemiptera (s.l.)
C. Infraorders of Auchenorrhyncha
D. The superfamilies and families of Fulgoromorpha
E. Diversity of planthoppers (in brief)
F. Phylogeny of the (extant) planthopper families
(the sections noted below are planned but incomplete)
G.Changes in planthopper family-level classification since the Metcalf catalog (planned)
H. Phylogeny and Higher Classification within Families (links to new pages)
I. References
(don’t know how to make section jumps)
This is an overview of the higher classification of planthoppers beginning with ordinal classification. At present the topic is presented in a simplistic way. Formal phylogenetic analyses (i.e., data driven, using computer algorithms, resulting in repeatable analyses) will be emphasized, when available.
A. Ordinal Classification (order Hemiptera)
As originally conceived (i.e., by Linnaeus 1758), the order Hemiptera was broadly defined to include the true bugs (now Heteroptera), scales, aphids, psyllids and allies (now Sternorrhyncha), true hoppers (now Auchenorrhyncha), and thrips (now Thysanoptera). The groupings “Heteroptera” and “Homoptera” (within Hemiptera) were first introduced by Latreille (1810a, b). By the early part of the 20th century, most workers defined these groups as two as independent orders – the true bugs as the order Hemiptera in the strict sense (i.e., the Heteroptera) and the true hoppers, scales and allies as the order Homoptera (e.g., Borrer et al. 1981). This is the classification that most North American entomologists learned (i.e., the first 6 editions of Borrer et al’s An Introduction to the Study of Insects, 1954-1981).
In the latter half of the 20th century, a series of studies challenged the general concept of maintaining Hemiptera and Homoptera as separate orders. Morphological investigations – including Goodchild (1966), Schlee (1969), Bourgoin (1986a, b; 1993); and Sweet (1996) – provided varied lines of evidence that cast doubt on the two order arrangement. However, it was the molecular evidence (Cambell et al. 1995, Sorensen et al. 1995, von Dohlen & Moran 1995) that most strongly challenged the two order arrangement. On the basis of 18s rDNA sequences, Cambell et al. (1995) and Sorensen et al. (1995) showed that the Heteroptera was derived within the Homoptera.
Here are the trees from Campell et al. (1995):

Similarly (and independently) von Dohlen and Moran (1995) (also based on 18S rDNA) found a paraphyletic Homoptera as follows:

Additionally, Sorenson et al. (1995) argued not only that the molecular evidence did not support monophyly of the Homoptera, but recent morphological and paleontological evidence did not either. Subsequent investigations (summarized, e.g., in Bourgoin & Campbell 2002) have supported the premise that the Heteroptera are derived within the “Homoptera” (see, e.g., Misof et al. 2014). The combined grouping ” Heteroptera + Homoptera” is logically termed Hemiptera (although this still confuses some who learned “Hemiptera” in the sense of Heteroptera). “Homoptera” is not a valid taxon and should be abandoned.
B. The Suborders of Hemiptera (s.l.) (with special reference to Auchenorrhyncha)
A result of the work of Sorensen et al. (1995) was that the traditional homopteran suborder ‘Auchenorrhyncha’ was found to be paraphyletic (or at least supporting evidence was not found for auchenorrhynchan monophyly). In contrast, the other groups – the traditional suborders Sternorrhyncha, Heteroptera, and Coleorrhyncha (the moss bugs, wiki, species file) were monophyletic (there was prior evidence that the Coleorrhyncha – traditionally a suborder of “Homoptera” – was actually the sister group to Heteroptera, e.g., Schlee 1969, Hamilton 1981, Wheeler et al. 1993). This arrangement suggested to Sorensen et al. (1995) four subordinal groupings – the sternorrhynchs, the planthoppers (fulgoromorphs), the cicadomorphs, and the Coleorrhyncha + Heteroptera. To these groups, they applied a new nomenclature, conforming the subordinal names using a -rrhyncha suffex; these were the Sternorrhyncha (retained in the usual sense), Clypeorrhyncha (‘shield-nose’) for extant, monophyletyic cicadomorphs; Archaeorrhyncha (‘ancient-nose’) for extant fulgoromorphs; and Prosorrhyncha (‘front’ or ‘forward-nose’) for Heteropteroidea – the combined grouping of Coleorrhyncha + Heteroptera. While an initial concern was that the ‘new’ groupings would not be exactly the same as the traditional higher taxa (hence the need for a new nomenclature), it subsequently became apparent that the new nomenclature conformed exactly to the old concepts of Sternorrhyncha, Heteroptera, Coleorrhyncha, Fulgoromorpha and Cicadomorpha; and Bourgoin & Campbell (2002) recommended discontinuing use of the new terminology.
The problem of Auchenorrhynchan monophyly (the ‘Auchenorrhyncha question’; Cryan & Urban 2011) proved to be difficult to resolve, and a series of studies (reviewed in Cryan & Urban 2011) suggested auchenorrhynchan paraphyly. However, Cryan & Urban (2011) produced strong evidence in support of the monophyly of Auchenorrnyncha. They used DNA nucleotide sequences from 7 gene regions (18S rDNA, 28S rDNA, histone H3, histone 2A, wingless, cytochrome c oxidase I and NADH dehydrogenase subunit 4), and found strong support (i.e., 100% Bayesian posterior probabilities) for the monophyly of the Auchenorrhyncha. While these results should settle the debate, recent mitogenomics studies have provided mixed results, although most of these studies were not intended to directly address the matter. However, the monophyly of Auchenorrhyncha has now been supported by several studies, including genomics (at least 3, but I will have to look up the references to add them here; this one is suggestive, at least and maybe this one, and this one).
The suborders of Hemiptera remain Sternorrhyncha, Auchenorrhyncha, Heteroptera and Coleorrhyncha (or Prosorrhyncha for the combined grouping), despite ongoing questions. (Note: some sources assert that Heteroptera and Homoptera are – or should be – treated as suborders of Hemiptera (e.g., wiki [as of Jan 2014]) but these data do not support this premise, and this arrangement has never been suggested in the context of phylogenetic research.)
Here is the tree from their results:

C. Infraorders of Auchenorrhyncha
Within the Auchenorrhyncha, 2 monophyletic lineages are recognized: the Fulgoromorpha (planthoppers) and Cicadomorpha (leafhoppers, treehoppers, cicadas [and here, here, here, here, &c] and spittlebugs) (e.g., Bourgoin et al. 1997, Bourgoin & Campbell 2002, Cryan & Urban 2011). These lineages are usually treated as infraorders (as opposed to their interim use as suborders by e.g., Bourgoin & Campbell 2002). However, some authors continue to assert that the monophyly of the Auchenorrhyncha has not been demonstrated to their satisfaction and so continue to use the Cicadomorpha and Fulgoromorpha as full suborders.
D. The Superfamilies and Families of Fulgoromorpha (the planthoppers)
The infraorder Fulgoromorpha is divided into three superfamilies (all taxon numbers from FLOW; Bourgoin 2014):
Superfamily Coleoscytoidea Martynov, 1935
1 extinct family (Coleoscytidae) (FLOW, fossilworks), 3 genera, 10 species (Upper Permian of the Russian Federation )
Superfamily Surijokocixioidea Shcherbakov, 2000
1 extinct family (Surijokocixiidae) (FLOW, fossilworks), 5 genera, 6 species (Upper Permian and Triassic, Russian Federation)
Coleoscyta and Surijokocixius from Bourgoin et al. (2004)


Generalized, partly hypothetical illustration of Surijokocixiidae (Shcherbakov 2000)

Superfamily Fulgoroidea Latreille 1807 (p. 163) (authorship is sometimes given as Duméril 1820: 509) (Metcalf 1947 cited Duméril 1820 as the first use of Fulgorelle, but specified authorship as Latreille) (Jurassic to present).
Some authors equivocate on the number of fulgoroid families – saying ‘about 20’. I will firmly state that there are currently 21 extant (and 9 extinct) recognized families of Fulgoroidea as follows († extinct):
E. Diversity of Planthoppers (in brief)
These numbers are from FLOW (this page) as of 13 Jan. 2019 (family links below to FLOW)
| Families | Genera | Species |
| Superfamily Coleoscytoidea | ||
| Coleoscytidae† | 3 | 10 |
| Superfamily Surijokocixioidea | ||
| Surijokocixiidae† | 5 | 6 |
| Subtotal | 8 | 16 |
| Superfamily Fulgoroidea (extant) | ||
| Acanaloniidae Amyot & Audinet-Serville, 1843 | 7 | 94 |
| Achilidae Stål, 1866 | 160 | 513 |
| Achilixiidae Muir, 1923 | 2 | 24 |
| Caliscelidae Amyot & Audinet-Serville, 1843 | 76 | 234 |
| Cixiidae Spinola, 1839 | 231 | 2498 |
| Delphacidae Leach, 1815 | 422 | 2212 |
| Derbidae Spinola, 1839 | 163 | 1686 |
| Dictyopharidae Spinola, 1839 | 156 | 733 |
| Eurybrachidae Stål, 1862 | 41 | 201 |
| Flatidae Spinola, 1839 | 292 | 1433 |
| Fulgoridae Latreille, 1807 | 143 | 765 |
| Gengidae Fennah, 1949 | 2 | 3 |
| Hypochthonellidae China & Fennah, 1952 | 1 | 1 |
| Issidae Spinola, 1839 | 200 | 1034 |
| Kinnaridae Muir, 1925 | 24 | 115 |
| Lophopidae Stål, 1866 | 48 | 173 |
| Meenoplidae Fieber, 1872 | 23 | 161 |
| Nogodinidae Melichar, 1898 | 95 | 368 |
| Ricaniidae Amyot & Serville, 1843 | 64 | 432 |
| Tettigometridae Germar, 1821 | 14 | 88 |
| Tropiduchidae Stål, 1866 | 186 | 660 |
| Subtotal | 2350 | 13428 |
| Superfamily Fulgoroidea (extinct) | ||
| Dorytocidae Emeljanov & Shcherbakov, 2018 | 1 | 1 |
| Fulgoridiidae† Handlirsch, 1939 | 17 | 130 |
| Lalacidae† Hamilton, 1990 | 9 | 17 |
| Jubisentidae† Zhang, Ren & Yao, 2019 | 2 | 2 |
| Mimarachnidae† Shcherbakov, 2007a | 7 | 7 |
| Neazoniidae† Szwedo, 2007 | 1 | 3 |
| Perforissidae† Shcherbakov, 2007b | 7 | 8 |
| Qiyangiricaniidae† Szwedo, Wang & Zhang, 2011 | 1 | 1 |
| Weiwoboidae† Lin, Szwedo, Huang & Stroinski, 2010 | 1 | 1 |
| Subtotal | 46 | 170 |
| Total | 2402 | 13614 |
F. Phylogeny of the (extant) planthopper families
An early cladistic consideration of the phylogeny of the extant planthoppers (based on morphology) was that of Asche (1987), who essentially postulated that the Tettigometridae were basal, followed by Cixiidae + Delphacidae and a clade consisting of all other extant families.
Phylogeny from Asche (1987) (left); same (right) with positions of other families inferred from text (from Urban & Cryan 2007)

Asche (1987) noted that the Cixiidae + Delphacidae possess an ‘orthopteroid’ ovipositor (modified in the advanced lineages), and fused gonapophyses on abdominal segment 9. He also noted that subterranean nymphs are plesiomorphic, and that Cixiidae lacked any convincing apomorphies.
Soon after, Emeljanov (1991) discussed the morphological features of the planthopper families and provided the following tree:

Wilson et al. (1994) subsequently summarized the planthopper phylogeny as:

The first substantive molecular examination of the phylogeny of planthoppers was that of Bourgoin et al. (1997) who used 18S rDNA to examine whether the Tettigometridae were a basal taxon, sister to the rest of the Fulgoroidea (as had been supposed by Asche 1987 and Emeljanov 1991), or were more advanced. They found that Tettigometridae was an advanced lineage, for example:
Tree from Bourgoin et al. (1997) using 18S rDNA:
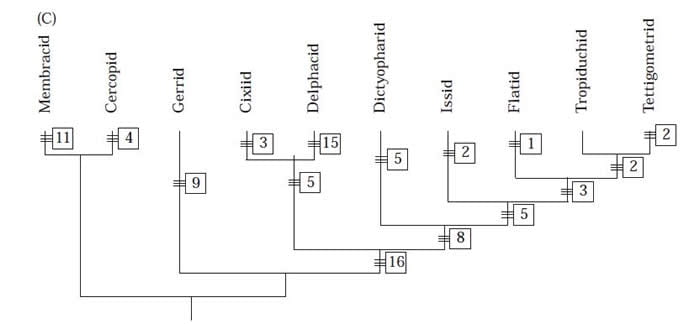
Additional phylogenetic work on planthopper was undertaken by Yang & Fang (1993) (based on nymphal morphology), Yeh & Yang (1999) (based on 28S rDNA), Yeh et al. (1999) (based on 16S rDNA). Bourgoin and Campbell (2002) summarized Hemipteran phylogeny evidence to that point as follows

Subsequently Yeh et al. (2005) analyzed 16S rDNA using neighbor-joining and minimum evolution techniques, producing several hypotheses, e.g.,

(A neighbor-joining tree based on the Kimura-2-parameter distance model)
Possibly the most interesting result from Yeh et al. (2005) is that Issidae (s.l.) was not monophyletic (viz. Caliscelinae, Tonginae, and Hemisphaerinae did not group with Issidae (Issinae).
The first molecular phylogeny using multiple genes and with most families represented was that of Urban & Cryan (2007). They presented phylogenetic reconstructions based on DNA nucleotide sequence data from four loci (18S rDNA, 28S rDNA, Histone 3 and Wingless) analyzed using both maximum parsimony and partitioned Bayesian criteria for 71 planthopper taxa in 19 families (lacking Gengidae and Hypochthonellidae).
The Bayesian 50% consensus tree looks like this:

Summarized this way:
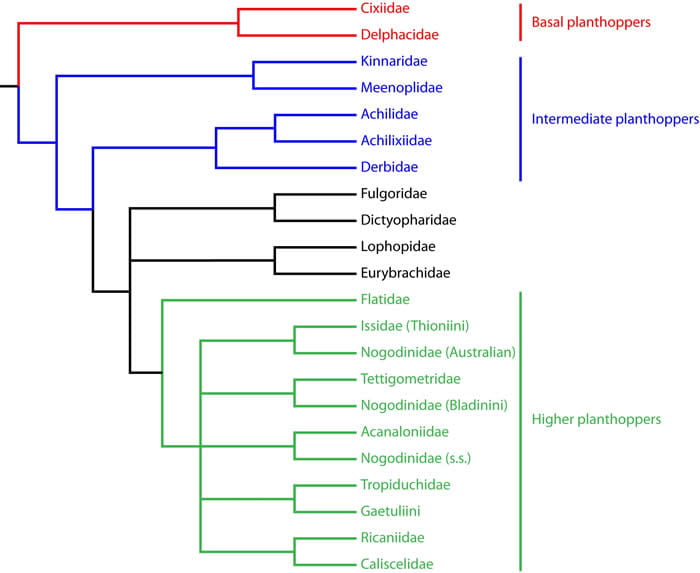
Based on this phylogenetic work, as well as that preceding it, several inferences can be made:
1. Of the extant planthopper lineages, the Cixiidae and Delphacidae are basal (the ‘basal planthoppers’). The relationship between the Cixiidae and Delphacidae is unclear; the families may be monophyletic or the Delphacidae may be derived within the Cixiidae.
2. The Kinnaridae, Meeneplidae, Achilidae, Achilixiidae and Derbidae are (probably) intermediate planthoppers (advanced relative to Cixiidae + Delphacidae, but not near the tip of the tree). Each are probably monophyletic (the placement of Amblycratus above is an artifact), but that is not certain. Some long-branch issues complicate analyses. It has subsequently been more clearly recognized (e.g., Urban & Cryan 2009), that lineages of planthoppers may evolve at different rates. This implies that phylogenetic methods that are more sensitive to variations in molecular evolutionary rate (e.g., maximum parsimony) are less suitable than methods that are less sensitive to molecular rate variation e.g., Bayesian). Unfortunately, the later is much more computationally intensive, and time consuming (days or weeks for analytical results), than the former.
3. The Fulgoridae + Dictyopharidae are sister groups that (usually) place above the intermediate lineages, but not among the higher planthoppers.
4. Likewise, the Eurybrachidae + Lophopidae are sister-groups, usually placed among the higher planthoppers.
5. Tettigometridae is advanced (among the higher planthoppers), but placement of the family is problematic because of long-branch issues.
6. The Flatidae, Issidae, Tettigometridae, Nogodinidae, Acanaloniidae, Tropiduchidae, Ricaniidae, Caliscelidae (and presumably Gengidae and Hypochthonellidae) are advanced lineages (constituting the ‘higher planthoppers’). The relationship among these families is not entirely clear.
7. Nogodinidae as currently comprised is paraphyletic. Other families of higher planthoppers may also be paraphyletic (or at least, may not be properly composed and should not be assumed to be monophyletic).
8. Supporting the contention of Emeljanov (1999), Caliscelidae and Acanaloniidae are families (not subfamilies of Issidae).
9. Additional phylogenetic work is needed with sampling at the subfamily (or tribe) level, and probably including more genes.
As an aside, the diagnostic feature (used in many planthopper family keys) of having many (a row) or few (2, 0) teeth on the second tarsomere of the hind leg maps on this tree nicely; all the basal taxa up to and including Fulgoridae + Dictyopharidae have a row of black-tipped teeth (B, below), and all the derived lineages (Lophopidae+Eurybrachidae and all higher planthoppers) have reduced spinulation (A, below).
Second tarsomere of the hind leg of Acanaloniidae (A) and Cixiidae (B)

Finally, a more recent molecular phylogeny was presented by Song & Liang (2013; which they called ‘preliminary’). They expanded upon the work of Urban & Cryan (2007) with increased taxon sampling (133 species from 110 genera in 19 families) and new genetic loci (but still used 4 genes). They used nuclear (18S and 28S) and mitochondrial (16S and cytb) markers, and subjected these data to maximum likelihood and Bayesian analyses. They also estimated divergence times using Beast.
Here is the Maximum Likelihood tree from Song & Liang (2013) (their Bayesian tree [link to jpg] has a peculiar branching pattern, making it not compelling).
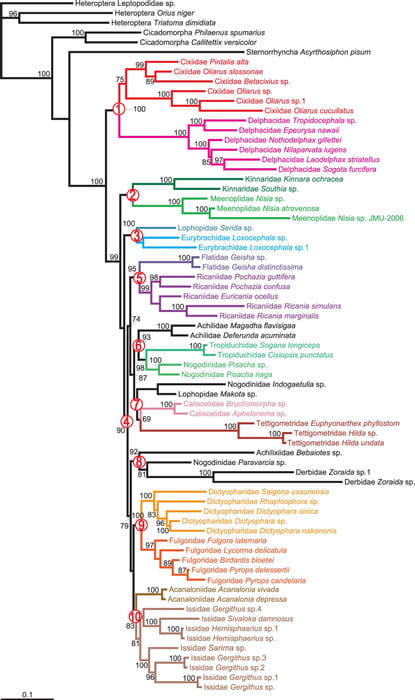
Song and Liang (2013) (their Figure 1.) Maximum-likelihood tree estimated from the combined datatset of 65 taxa of Fulgoroidea. Values above branches denote ML bootstrap support (≥70). The red numbers in circles correspond to ten major clades discussed in their text.
The results of Song and Liang (2013) are similar to Urban and Cryan (2007) in that
1. Cixiidae & Delphacidae are basal.
2. Kinnaridae + Meenoplidae are intermediate.
3. Dictyopharidae + Fulgoridae are sister groups.
4. Eurybrachidae and Lophopidae are sister groups.
5. Nogodinidae is paraphyletic.
6. Derbidae, Achilixiidae and Tettigometridae have long branches.
7. The ‘higher planthoppers’ are derived.
8. Caliscelidae does not group with Issidae (supporting Emeljanov, 1999)
These results differ from Urban & Cryan (2007) by:
1. The intermediate planthopper lineages of Achilidae, Achilixiidae and Derbidae are interspersed among advanced lineages.
2. The relationships among the higher planthoppers differ from Urban & Cryan.
The results of Song & Liang’s (2013) divergence time estimation are also interesting

Song and Liang (2013) (their Figure 3). Chronogram of Fulgoroidea estimated using the Bayesian phylogenetic software BEAST. Time units are in millions of years before present. Estimated divergence times are shown near nodes, while blue bars represent 95% credibility intervals. The values on the branches denote the posterior probabilities (≥0.9). (doi:10.1371/journal.pone.0058400.g003)
Still, so much more needs to be done, and there have been no more published molecular investigations of planthopper families since then; however, there have been several more limited phylogenetic investigations, which I still need to summarize here.
G. Changes in planthopper family-level classification since the Metcalf catalog (planned)
H. Phylogeny and Higher Classification within Families
(links below go to a description of the higher classification of that family)
Acanaloniidae Amyot & Serville, 1843 (link to home page of this site which discusses classification)
Achilidae Stål, 1866
Achilixiidae Muir 1923
Caliscelidae Amyot & Serville, 1843
Cixiidae Spinola, 1839
Delphacidae Leach, 1815
Derbidae Spinola, 1839
Dictyopharidae Spinola, 1839
Eurybrachidae Stål, 1862
Flatidae Spinola, 1839
Fulgoridae Latreille 1807
Gengidae Fennah, 1949
Hypochthonellidae China & Fennah, 1952
Issidae Spinola, 1839
Kinnaridae Muir, 1925
Lophopidae Stål, 1866
Meenoplidae Fieber, 1872
Nogodinidae Melichar, 1898
Ricaniidae Amyot & Serville, 1843
Tettigometridae Germar, 1821
Tropiduchidae Stål, 1866
I. References
Amyot, C. J. B. and J. G. Serville. 1843. Deuxième partie. Homoptères. Homoptera Latr. Histoire Naturelle des insectes. Hemiptères 1843: 1-676.
Asche, M. 1988. Preliminary thoughts on the phylogeny of Fulgoromorpha (Homoptera, Auchenorrhyncha). Pp. 47-53. In: Proceedings of the 6th Auchenorrhyncha Meeting, Turin, Italy, September 7-11, 1987.
Borror, D. J., D. M. De Long and C. A. Triplehorn. 1981. An Introduction to the Study of Insects, Fifth Edition. Saunders College Publishing.
Bourgoin, T. 1986a. Morphologie imaginale du tentorium des Hemiptera Fulgoromorpha. International Journal of Insect Morphology and Embryology 15(4): 237-252.
Bourgoin, T. 1986b. Valeur morphologique de la lame maxillaire chez les Hemiptera; remarques phylogénétiques. Annales de la Société Entomologique de France 22(4): 413-422.
Bourgoin, T. 1993. Female genitalia in Fulgoromorpha Hemiptera Fulgoromorpha morphological and phylogenetical data. Annales de la Société Entomologique de France (Nouvelle Serie) 29(3): 225-244.
Bourgoin, T. 2014. FLOW (Fulgoromorpha Lists on The Web): a world knowledge base dedicated to Fulgoromorpha. Version 8, updated Jan. 14, 2014; accessed Jan 16,2014. http://hemiptera-databases.org/flow/
Bourgoin, T. and B. C. Campbell. 2002. Inferring a phylogeny for Hemiptera: falling into the ‘Autapomorphic Trap’. Denisia 4: 67-82. (pdf)
Bourgoin, T., J. D. Steffen-Campbell and B. C. Campbell. 1997. Molecular phylogeny of Fulgoromorpha (Insecta, Hemiptera, Archaeorrhyncha). The enigmatic Tettigometridae: evolutionary affiliations and historical biogeography. Cladistics 13: 207-224.
Campbell, B. C., J. D. Steffen-Campbell, J. T. Sorensen, and R. J. Gill. 1995. Paraphyly of Homoptera and Auchenorrhyncha inferred from 18S rDNA nucleotide sequences. Systematic Entomology 20: 175-194.
China, W. E. and R. G. Fennah. 1952. On a remarkable genus of Fulgoroid Homoptera representing a new family. Annals and Magazine of Natural History. London. (Ser. 12) 5: 189-199.
Cryan, J. R. and J. M. Urban. 2011. Higher-level phylogeny of the insect order Hemiptera: is Auchenorrhyncha really paraphyletic? Systematic Entomology 37(1): 7-21.
von Dohlen, C. D. and N. A. Moran. 1995. Molecular Phylogeny of the Homoptera: A Paraphyletic Taxon. Journal of Molecular Evolution 41: 211-223.
Duméril, A. M. C. 1820b. Fulgore, Fulgora. In Dictionnaire des sciences naturelles, dans lequel on traite méthodiquement des différens êtres de la nature, considérés soit en eux-mêmes, d’après l’état actuel de nos connoissances, soit relativement à l’utilité qu’en peuvent retirer … et les artes. F. G. Levrault, Strasbourg. 17: 507-509.
Emeljanov, A. F. 1991. An attempt to construct a phylogenetic tree for planthoppers (Homoptera, Cicadina). Entomological Review 70(1): 24-28.
Emeljanov, A. F. 1999. Notes on delimitation of families of the Issidae group with description of a new species of Caliscelidae belonging to a new genus and tribe (Homoptera, Fulgoroidea). Zoosystematica Rossica 8(1): 61-72.
Emeljanov A. F. and D. E. Shcherbakov. 2018. The longest-nosed Mesozoic Fulgoroidea (Homoptera): a new family from mid-Cretaceous burmese amber. Far Eastern Entomologist 354: 1-14
Fennah, R. G. 1949. A new genus of Fulgoroidea (Homoptera) from South Africa. Annals and Magazine of Natural History. London. (Ser. 12) 2: 111-120.
Fieber, F. X. 1872. Katalog der europäischen Cicadinen, nach Originalien mit Benützung der neuesten Literatur. Druk und Verlag von Carl Gerold’s Sohn, Wien (Austria). 19 pp.
Germar, E. F. 1821. Bemerkungen über einige Gattungen der Cicadarien. Magazin der Entomologie 4: 1-106.
Goodchild, A.J.P. 1966. Evolution of the ailmentary canal in the Hemiptera. Biological Review 41: 97-140.
Hamilton, K.G.A. 1981. Morphology and evolution of the Rhynchotan head (Insecta: Hemiptera, Homoptera). Canadian Entomologist 113: 953-974.
Hamilton, K.G.A. 1990. Chapter 6. Homoptera. In Grimaldi, D.A. (ed.). Insecta from the Santana formation, lower Cretaceous, of Brazil. Bulletin of the American Museum of Natural History 195: 82-122.
Handlirsch, A. 1939. Neue Untersuchungen über die fossilen Insekten, Teil 2. Annalen des Naturhistorischen Museums in Wien 49: 1-240.
Latreille, P. A. 1807. Sectio secunda. Familia quarta. Cicadariae. Cicadaires. Genera Crustaceorum et Insectorum secundum ordinem naturalem in familias disposita, iconibus exemplisque plurimis explicata 3: 1-258.
Latreille, P. A. 1810a. Section Premiere. Hètèroptères. Heteroptera. and Section II. Homoptères. Homoptera. Pp. 254-266. In Considérations générales sur l’ordre naturel des animaux composant les classes des Crustacès, des Arachnides, et des Insectes; avec un tableau méthodique de leurs genres, disposés en familles. F. Schoell,. Paris. Pp. 1-420.
Latreille, P. A. 1810b. Table des genres avec l’indication de l’espèce qui leur sert de type. In Considérations générales sur l’ordre naturel des animaux composant les classes des Crustacès, des Arachnides, et des Insectes; avec un tableau méthodique de leurs genres, disposés en familles. F. Schoell, Paris. Pp. 421-444.
Leach, W. E. 1815. Entomology. The Edinburgh Encyclopedia 9: 57-172.
Lin Q., Szwedo J., Huang D. & Stroinski A. 2010. Weiwoboidae fam. nov. of ‘Higher’ Fulgoroidea (Hemiptera: Fulgoromorpha) from the Eocene deposits of Yunnan, China. Acta Geological Sinica (English Edition) 84(4): 751-755.
Linné, C. 1758a. II. Hemiptera. In Systema naturae: per regna tria naturae, secundum classes, ordines, genera, species cum characteribus, differentiis, synonymis, locis. Editio decima, reformata. L. Salvii . [Stock]holmiae. 1: 1-824.
Martynov, A. V. 1935. Permian fossil insects from Arkhangelsk District. Part.5. Homoptera. Trudy Paleozoologischeskogo Instituta Akademii Nauk SSSR 4: 1-35.
Melichar, L. 1898. Monographie der Ricaniiden (Homoptera). Annalen des k.k Naturhistorischen Hofmuseums 13: 197-359.
Metcalf, Z. P. 1947. General Catalogue of the Homoptera. Fascicle IV Fulgoroidea. Part 9 Fulgoridae. Smith College, Northhampton, Massachusetts.
Misof et al., 2014. Phylogenomics resolves the timing and pattern of insect evolution. Science 346: 763-767.
Mozaffarian, F., T. Bourgoin and M. R. Wilson. 2018 Nomenclatural changes in the higher classification of the family Tettigometridae (Hemiptera: Fulgoroidea) with description of a new tribe and new species and a review of the Iranian tettigometrid fauna. Zootaxa 4392 (3): 469–490 doi: 10.11646/zootaxa.4392.3.3.
Muir, F. A. G. 1923. Achilixius, a new genus, constituting a new family of the Fulgoroidea (Homoptera). Philippine Journal of Science. Manila 22: 483-487.
Muir, F. A. G. 1925. On the genera of Cixiidae, Meenoplidae and Kinnaridae (Fulgoroidea, Homoptera). Pan-Pacific Entomologist 1: 97-110.
Schlee, D. 1969. Morphologie und Symbiose, ihre Beweiskraft für die Verwandtschaftsbeziehungen der Coleorrhyncha (Insecta, Hemiptera): phylogenetische Studien an Hemiptera IV: Heteropteroidea (Heteroptera+ Coleorrhyncha) als monophyletische Gruppe. Staatliches Museum für Naturkunde 210: 1–27.
Shcherbakov, D. E. 2000. Permian faunas of Homoptera (Hemiptera) in relation to phytogeography and the Permo-Triassic crisis. Supplement N°3S. Paleontological Journal 3: 251-267.
Shcherbakov, D. E. 2007a. Mesozoic spider mimics – Cretaceous Mimarachnidae fam. n. (Homoptera: Fulgoroidea). Russian Entomological Journal 16(3): 259-264.
Shcherbakov, D. E. 2007b. An extraordinary new family of Cretaceous planthoppers (Homoptera: Fulgoroidea). Russian Entomological Journal 16(2): 139-154.
Song, N. and Ai-Ping Liang. 2013. A Preliminary Molecular Phylogeny of Planthoppers (Hemiptera: Fulgoroidea) Based on Nuclear and Mitochondrial DNA Sequences. PLoS one DOI: 10.1371/journal.pone.0058400
Spinola, M. 1839. Essai sur les Fulgorelles, sous-tribu de la tribu des Cicadaires, ordre des Rhyngotes. Annales de la Société Entomologique de France. Paris 8: 133-337.
Sorensen, J. T., B. C. Campbell, R. J. Gill and J. D. Steffen-Campbell. 1995. Non-monophyly of Auchenorrhyncha (“Homoptera”), based upon 18S rDNA phylogeny: eco-evolutionary and cladistic implications within pre-heteropterodea Hemiptera (s.l.) and a proposal for new monophyletic suborders. Pan-Pacific Entomologist 71(1): 31-60.
Stål, C. 1866. Hemiptera Homoptera Latr. Hemiptera Africana 4: 1-276.
Stål, C. 1862. Synonymiska och systematiska anteckningar öfver Hemiptera. Ofversigt af Kongliga Svenska Vetenskaps-Akademiens Förhandlingar 19: 479-504.
Sweet, M. H. 1996. Comparative external morphology of the pregenital abdomen of the Hemiptera. Pp. 119-158. In C. W.Schaefer, (ed.). Studies on Hemipteran Phylogeny. Thomas Say Publications in Entomology: Proceedings.
Szwedo, J., T. Bourgoin, and F. LeFèbvre (eds.). 2004. Fossil Planthoppers (Hemiptera: Fulgoromorpha) of the world. An annotated catalogue with notes on Hemiptera classification. Studio 1, Warsaw, Poland.
Szwedo, J. 2007. Nymphs of a new family Neazoniidae fam. n. (Hemiptera: Fulgoromorpha: Fulgoroidea) from the Lower Cretaceous Lebanese amber. African Invertebrates 48(1): 127-143.
Szwedo, J., Wang Bo and Zhang Haichun. 2011. An extraordinary early Jurassic planthopper from Hunan (China) representing a new family Qiyangiricaniidae fam. nov. (Hemiptera: Fulgoromorpha: Fulgoroidea). Acta Geological Sinica (English Edition) 85(4): 739-748.
Urban, J. M. and J. R. Cryan. 2007. Evolution of the planthoppers (Insecta: Hemiptera: Fulgoroidea). Molecular Phylogenetics and Evolution 42(2): 556-572.
Urban J. M. and J. R. Cryan. 2009. Entomologically famous, evolutionarily unexplored: The first phylogeny of the lanternfly family Fulgoridae (Insecta: Hemiptera: Fulgoroidea). Molecular Phylogenetics and Evolution 50: 471-484.
Wheeler, W. C., R. T. Schuh and R. Bang. 1993. Cladistic relationships among higher groups of Heteroptera: congruence between morphological and molecular datasets. Entomologica Scandinavica 24: 121-137. (pdf, not a good scan)
Wilson, S. W., C. Mitter, R. F. Denno, and M. R. Wilson. 1994. Evolutionary patterns of host plant use by delphacid planthoppers and their relatives. In: R. F. Denno and T. J. Perfect, (eds.). Planthoppers: Their Ecology and Management. Chapman and Hall, New York. Pp. 7-45 & Appendix.
Yang, C. T. and S. J. Fang. 1993. Phylogeny of Fulgoromorpha nymphs, first results. Pp. 25-26. In: Proceedings. 8th Auchenorrhyncha Congress, Delphi, Greece, 9-13 Aug. 1993.
Yeh, W.B., and C.T. Yang. 1999. Fulgoromorpha phylogeny base on 28S rDNA nucleotide sequence. Chinese Journal of Entomology (Suppl.) 11: 87-111.
Yeh, W. B., C. T. Yang, and C. F. Hui. 1999. A molecular phylogeny of planthoppers (Homoptera: Fulgoroidea) inferred from mitochondrial 16S rDNA sequences. Pp. 57-82. In: Proceeding of the Symposium of Insect Systematics and Evolution. Department Entomology National Taiwan University and Taiwan Museum. Taipei, Taiwan. [in Chinese]
Yeh, W. B., C. T. Yang, and C. F. Hui. 2005. A molecular phylogeny of planthoppers (Hemiptera: Fulgoroidea) inferred from mitochondrial 16S rDNA sequences. Zoological Studies 44(4): 519-535. (PDF).
Zhang. Xiao, Dong Ren and Yunzhi Yao. 2019. A new family Jubisentidae fam. nov. (Hemiptera: Fulgoromorpha: Fulgoroidea) from the mid-Cretaceous Burmese amber. Cretaceous Research 94: 1-7

