[Back to North American Cixiidae]
[Return to Planthoppers of North America]
Contents
Family Cixiidae Spinola, 1839
Subfamily Cixiinae Spinola, 1839
Tribe Oecleini Muir, 1922
Genus: Oecleus Stål, 1862.
Type species (in original combination): Oecleus seminiger Stål, 1862.
Synonyms
None.
Distribution
Widespread in New World, most species in the southwestern US and Mexico.
Recognized species
There are 65 species as follows
1 Oecleus arnellus Ball & Klingenberg, 1935 – USA: AZ, NV, TX, UT
2 Oecleus augur Kramer, 1977 – USA: AZ, TX
3 Oecleus balli Kramer, 1977 – USA: AZ, CA, NV
4 Oecleus biflagellatus O’Brien, 1982a – USA: AZ
5 Oecleus borealis Van Duzee, 1912a – USA: AL, AZ, CT, DC, FL, GA, IL, KS, MD, MI, MS, NC, NJ, NY, OH, OK, PA, SC, TX, UT, VA; CAN: ON
= Oecleus bilineatus Caldwell, 1938b: 305; syn. by Kramer 1977: 433-434.
6 Oecleus campestris Ball, 1902a – USA: AZ, CA, CO, NM, TX (reported in error: USA: CT, MS)
7 Oecleus capitulatus Van Duzee, 1912a – USA: AZ
= Oecleus triplicatus Ball & Klingenberg, 1935: 198; syn. by Kramer 1977: 420, 422.
8 Oecleus centronus Ball & Klingenberg, 1935 – USA: AZ
9 Oecleus chrisjohni Kramer, 1977 – USA: IL, MO, TX
10 Oecleus cucullus Kramer, 1977 – USA: AZ, CA, TX, UT
11 Oecleus decens Stål, 1862a – Mexico (Baja California, Jalisco, Morelos, Sonora) (reported in error: USA: CA, CT, DC, LA, NC, NJ, NM, TX)
12 Oecleus epetrion Kramer, 1977 – USA: IL, TX
13 Oecleus excavatus Ball, 1902a – USA: AZ, CA, CO, NE, NM. OK
14 Oecleus fulvidorsum Ball, 1902a – USA: AZ, CA, CO, TX, UT
15 Oecleus glochin Kramer, 1977 – USA: CA, NV, UT
16 Oecleus jenniferae Kramer, 1977 – USA: AZ
17 Oecleus lineatus Ball, 1902a – USA: AZ, CA, NV, TX
18 Oecleus lyra Kramer, 1977 – USA: AZ
19 Oecleus martharum Kramer, 1977 – USA: AZ, CA, NV, UT
20 Oecleus monilipennis Van Duzee, 1923a – USA: CA, NM, NV, TX; Mexico (Baja California, Baja California sur; Gulf of California: Ceralbo, Espiritu Santo, Mejia, San Esteban and Santa Cruz Islands)
21 Oecleus natatorius Ball, 1937 – USA: AZ
22 Oecleus netrion Kramer, 1977 – USA: CA
23 Oecleus netron Kramer, 1977 – USA: AZ
24 Oecleus nolinus Ball & Klingenberg, 1935 – USA: AZ, NM, TX
= Oecleus texanus Ball, 1937: 181; syn. by Kramer 1977: 408, 410.
25 Oecleus obrieni O’Brien, 1982a – USA: TX
26 Oecleus obtusus Ball, 1902a – USA: AZ, CA, CO, ID, KS?, NE?, NV, OR, TX, UT, WA; CAN: BC; Mexico (Baja California, Baja California Sur)
27 Oecleus palton Kramer, 1977 – USA: AZ
28 Oecleus patulus Kramer, 1977 – USA: AZ
29 Oecleus pellucens Fowler, 1904: 90 – USA: AZ; Mexico (Chiapas, Federal District, Guerrero, Jalisco, Mexico, Michoacán, Oaxaca, Puebla, Quintana Roo, San Luis Potosí, Tabasco, Veracruz); El Salvador, Guatemala
30 Oecleus perpictus Van Duzee, 1929c – USA: AZ, CA Mexico (Baja California, Sonora)
31 Oecleus pigmy Ball & Klingenberg, 1935 – USA: AZ, CA, ID, NM, NV, TX, UT
32 Oecleus piperatus Ball & Klingenberg, 1935 – USA: AZ, CA; Mexico (Baja California)
33 Oecleus planus Ball & Klingenberg, 1935 – USA: CA, NV
34 Oecleus pontifex Kramer, 1977 – USA: AZ
35 Oecleus productus Metcalf, 1923 – USA: AZ, CA, DE, FL, GA, IL, MD, MO, MS, NC, NE, NM, NV, TX, UT; Mexico (Sonora)
36 Oecleus quadrilineatus Van Duzee, 1912a – USA: AZ
37 Oecleus rhion Kramer, 1977 – USA: AZ
38 Oecleus sagittanus Ball & Klingenberg, 1935 – USA: CA, NV, UT
39 Oecleus snowi Ball, 1905 – USA: AZ, CA, TX; Mexico (Baja California, Baja California Sur, Sonora)
41 Oecleus subreflexus Van Duzee, 1925 – USA: AZ, CA, NV, UT
= Oecleus cabazonus Ball & Klingenberg, 1935: 201; syn. by Kramer 1977: 404, 406.
= Oecleus pulchellatus Ball & Klingenberg, 1935: 201; syn. by Kramer 1977: 404, 406.
41 Oecleus tamiamus Ball & Klingenberg, 1935 -USA: FL, GA
42 Oecleus troxanon Kramer, 1977 – USA: UT
43 Oecleus vates Kramer, 1977 – USA: AZ
44 Oecleus venosus Van Duzee, 1912a – USA: CA, NV, OR, TX; CAN: BC
Neotropical species
1 Oecleus acutus Ball 1902:157 – Haiti
2 Oecleus addendus Fowler 1904: 91 – Guatemala
3 Oecleus apicatus Caldwell 1944:176 – Mexico (San Luis Potosí)
4 Oecleus apterapunctatus Caldwell 1944:198 – Mexico (Oaxaca)
5 Oecleus brunneus Fowler 1904: 91 – Mexico (Veracruz)
6 Oecleus cephalicus Caldwell 1944:200 – El Salvador
7 Oecleus concinnus Fowler 1904: 91 – Mexico (Guerrero)
8 Oecleus constrictus Caldwell 1944:199 – Mexico (Michoacán)
Oecleus decens Stål 1862:307 – Mexico (Baja California, Jalisco, Morelos, Sonora) (reported in error: USA: CA, CT, DC, LA, NC, NJ, NM, TX) (counted above)
9 Oecleus delongi Caldwell 1944:100 – Mexico (San Luis Potosí)
10 Oecleus dormido Bahder & Bartlett, 2022 – Coasta Rica
11 Oecleus dubius Caldwell 1944:175 – Guatemala
12 Oecleus infuscatus Caldwell 1944:198 – Mexico
13 Oecleus kasparyani Emelyanov 2007:291 – Mexico Northeast (Nuevo León)
14 Oecleus mackaspringi Bahder & Bartlett, 2019 – Jamaica
15 Oecleus minimus Fowler 1904: 90 – Mexico (Guerrero)
16 Oecleus monilipenis Van Duzee 1923:190 – Mexico, (Baja California)
17 Oecleus parallelus Caldwell 1944:176 – Mexico (Puebla)
Oecleus pellucens Fowler 1904: 91 – See above
18 Oecleus quinquilineatus Caldwell 1944:200 – Mexico (Guerrero, Morelos, Veracruz)
19 Oecleus seminiger Stal 1862:307 – Mexico (Veracruz)
20 Oecleus spatulatus Caldwell 1944:200 – Guatemala
21 Oecleus sergipensis Bartlett, Dos Passos, Gonçalves da Silva, Diniz & Dollet, 2018 (Brazil Northeast: Sergipe)
22 Oecleus teapae Fowler 1904: 89 – Mexico (Tabasco)
23 Oecleus tenellus Fowler, 1904: 89 – Mexico (Guerrero)
Economic Importance:
Limited, although Oecleus sergipensis was collected in association with coconut and may be explored as a possible pathogen vector.
Known plant associations:
(see also FLOW)
- Oecleus arnellus – Chrysothamnus speciosus Nutt. (Asteraceae)
- Oecleus balli – Atriplex (2 species; saltbush, Chenopodiaceae), Pluchea sericea (Nutt.) Coville (arrowweed, Asteraceae)
- Oecleus borealis – Chilopsis linearis (Cav.) Sweet (desert willow, Bignoniaceae)
- Oecleus campestris – Artemisia filifolia Torr. (sand sagebrush, Asteraceae)
- Oecleus centronus – Salicornia L. (pickleweed, Chenopodiaceae)
- Oecleus cucullus –Trixis californica Kellogg (American threefold, Asteraceae), Artemisia, Chilopsis linearis (Cav.) Sweet
- Oecleus fulvidorsum – Atriplex canescens (Pursh) Nutt. (fourwing saltbush, Chenopodiaceae)
- Oecleus glochin – Chrysothamnus, Hymenoclea Torr. & A. Gray ex A. Gray (burrobrush, Asteraceae)
- Oecleus lineatus – Pleiacanthus spinosus (Nutt.) Rydb. (thorn skeletonweed, Asteraceae, as Lygodesmia spinosa)
- Oecleus lyra – Dasylirion Zucc. (sotol, Liliaceae)
- Oecleus martharum – Atriplex (3 species),
- Oecleus monilipennis – Atriplex
- Oecleus natatorius – grasses
- Oecleus nolinus – Nolina Michx. (beargrass, Liliaceae), Yucca (Agavaceae)
- Oecleus obtusus – Pluchea (Asteraceae)
- Oecleus perpictus – Muhlenbergia porteri Scribn. ex Beal (bush muhly, Poaceae)
- Oecleus pygmy – Atriplex (3 spp.)
- Oecleus piperatus – Atriplex canescens (Pursh) Nutt. var. linearis (S. Watson) Munz (thinleaf fourwing saltbush as Atriplex linearis)
- Oecleus palnus – Ericameria paniculata (A. Gray) Rydb. (Mojave rabbitbrush as Chrysothamnus paniculatus (A. Gray) H.M. Hall)
- Oecleus productus – grasses, composites
- Oecleus sagittanus – Pluchea sericea (Nutt.) Coville (arrowweed, Asteraceae)
- Oecleus sergipensis – Cocos nucifera L. L., Phoenix sp. (Arecales, Arecaceae)
- Oecleus snowi – Baccharis salicifolia (Ruiz & Pav.) Pers. (mule-fat, Asteraceae as Baccharis glutunosa)
- Oecleus subreflexus – Atriplex lentiformis (Torr.) S. Watson (Big saltbush), Pluchea, Ambrosia (Ragweed, Asteraceae, as Franseria), Chrysothamnus
- Oecleus texanus – Yucca glauca Nutt. (soapweed yucca)
- Oecleus sp. – Cocos nucifera L. (coconut palm, Arecaceae)
- Oecleus sp. – Tamarix gallica L. (French tamarisk, Tamaricaceae)
Hosts from Wilson et al. 1994; plant names from USDA PLANTS or Tropicos.
Additional host records are recorded in FLOW here (some are dupes, others I’ll add above eventually).
Recognition:
Vertex compressed (carinae weak or obsolete), mesonotum with 5 conspicuous carinae; most taxa rather small.
U.S. species can be identified using Kramer (1977). Species identification is mostly based on male genitalia. The western richness makes identification challenging, but the few eastern species are less challenging.
Partial diagnosis from Kramer (1977)
” Small to moderately large cixiids (3.3-8.5 mm.); head in dorsal view (figs. 1-2), much narrower than pronotum with eyes large; vertex narrow, troughlike with sides and anterior margin carinate, sides parallel or subparallel, base narrowed and open or closed, apex variably produced beyond eyes; in lateral view (fig. 3), apex of head acutely or obtusely angled, lateral edges of vertex not more than slightly higher than upper margins of ventrally indented eyes, ocellus under each eye; in facial view sides and midline of frons and clypeus carinate, carina on midline of frons sometimes obsolete, frons elongated and narrowed toward vertex, clypeus triangular or subtriangular, ocellus on midline of frons just above fronto-clypeal suture; in dorsal view pronotum collarlike with irregular ridges, narrowest on midline, indented on posterior margin, declivant laterad, carinate or subcarinate on posterior and lateral margins; mesonotum longer on midline than combined lengths of vertex and pronotum, disc of mesonotum flattened and typically with five longitudinal carinae, central carina and lateral pair usually more distinct than pair flanking central carina, at times pair flanking central carina absent or reduced to pale pigmented lines, lateral portions of mesonotum declivant; antenna originating from large socket, scape reduced and collarlike, pedical round or quadrately rounded with some papillose sensoria, flagellum beadlike basally and filamentous distally; hind tibiae
without spines before apex; forewings essentially hyaline, rarely with color pattern, venation as in fig. 3, veins typically dotted with dark pustules which may or may not bear setae, pustules often highly obscure or obsolete.
Male genitalia: pygofer longest on ventral margin, anal flap elongated, paired slender symmetrical styles flanking median lobe of pygofer, styles and basal portion of asymmetrical aedeagus articulated by vertical connective, aedeagus transverse, consisting of shaft and distal reclinate flagellum, shaft with 1 to 3 processes, flagellum with 0 to 2 processes (see later discussion).
Generic diagnosis. Oecleus can be separated from all other Cixiidae in our fauna by characters of the head in dorsal view (figs. 1-2). The size of the eyes is exaggerated by the narrowness of the vertex. The vertex, even though often mainly parallel sided, is always narrower basally than apically and is separated from the face by a short transverse carina. The only genus in our fauna with which it might be confused is Oeclidius Van Duzee in the Kinnaridae. Oeclidius also has a narrow vertex but differs by having the vertex broadest basally, then tapered to a narrow or closed apex. Further, Oeclidius has a distinct carina on the midline of the pronotum; this character is absent in Oecleus. The male genitalia of the two genera differ in many respects; the most obvious difference is the lack of an aedeagal flagellum in Oeclidius.
Male genitalia. Successful separation of the species of Oecleus depends almost entirely upon the cryptic features of the male genital capsule. Figures 4-6 provide generalized views of these structures with the parts labeled according to the terminology used in this report. Those parts of the male genitalia of prime taxonomic value are the median lobe of the pygofer (MLP), anal flap (AF), and the various parts of the aedeagus (fig. 5). The aedeagus is the most complex component of the male structures. It consists of a sclerotized shaft and a recumbent semimembranous apical flagellum. At rest the flagellum is invariably found on the right of the shaft. In copulation the flagellum in flates and assumes a position at the apex of the shaft so that its sides are roughly parallel with those of the shaft. The gonopore is at the apex of the flagellum. Within the flagellum are two dif
ferently sclerotized oval structures which aid in the transfer of sperm. Basad of the flagellum, within the shaft on its long axis, the hollow middle rod (MR) provides the gonoduct. The number and shape of the processes found on the shaft and flagellum provide the most important taxonomic features. The number on the shaft may be 3, 2, or 1. When 3 are present, they are termed right process (RPS), middle process (MPS), and left process (LPS). When 2 are present, they are termed right process and left process. The flagellum may have 2, 1, or 0 processes. When 2 are present, they are called proximal process (PPF) and distal process (DPF). At times it is more useful to refer to the processes of the flagellum as inner process and outer process where they are contiguous, parallel, and subequal in length. Rarely the shaft bears an acute projection (APS) on the right
margin near the base or near the middle. With the exception of the median lobe of the pygofer (MLP) and the anal flap (AF), both of taxonomic value, the rest of the labeled parts in figs. 4-6 serve mainly to orient the reader.”

TEXT FIGS (From Kramer 1977). 1-3. Dorsal and lateral habitus views. Fig. 1, Oecleus perpictus Van Duzee. Fig. 2, Oecleus martharum Kramer, Fig. 3, Oecleus venosus Van Duzee.

Oecleus seminiger Stal from Fowler 1904 (the type species of the genus).
Key to species (males) modified from Kramer 1977
1- Shaft with acute projection on right side near base or middle (Fig. 10, below) … 2
1′- Shaft without such a projection … 3
2- Projection of shaft near base, median lobe of pygofer triangularly produced (fig. 7) … Oecleus rhion Kramer.
2- Projection of shaft near middle, median lobe of pygofer roundly produced (fig. 10) … Oecleus piperatus B&K
3- Shaft with one process (Fig. 15 [process from flagellum is not counted]) … 4
3′- Shaft with two or three processes … 5
4- Larger tawny species; face, carina included, tawny; forewings unmarked; anal flap in lateral view strongly convex on distal ventral margin (fig. 14) … Oecleus snowi Ball
4′- Smaller dark species; face, carina excepted, intense black; each forewing with dark, narrow, transverse, subapical stripe; anal flap in lateral view weakly convex on distal ventral margin (fig. 17) … Oecleus vates Kramer
Kramer 1977 Figs. 13-15 O. snowi, 16-18 O. vates.
5- Shaft with three processes … 6
5′- Shaft with two processes … 12
6- All processes more than half the length of the shaft, the apex of the middle process abruptly narrowed and hooked, nearly touching base in ventral view (fig. 19) … Oecleus martharum Kramer
6′- All processes not more than half the length of the shaft, the apex of the middle process gradually narrowed and curved to the right in ventral view (fig. 22) … 7
Kramer 1977 Figs. 19-21 O. martharum, 22-24 O. pigmy, 25-27 O. monilipennis.
7- Males less than 4 mm. long, usually about 3.5 mm … 8
7′- Males 4 mm. or longer … 10
8- Face, carina excepted, and all veins of forewings brown … Oecleus pigmy B&K (in part)
8′- Face pale or if brownish, carina excepted, then only veins beyond claval apex brown or partly brown … 9
9- Left process at apex turned to right, or right process straight or nearly so and not forming caliperlike structure with middle process in ventral view … Oecleus pigmy B&K (in part)
9′- Left process curved to left, and right process curved to form caliper like structure with middle process in ventral view … 10
10- Middle process [at apex of shaft, ventral view] stouter than right process in ventral view (fig. 25) … Oecleus monilipennis Van Duzee
10′- Middle process as stout or less stout than right process in ventral view … 11
11- Middle and right process subequal in stoutness in ventral view (fig. 28) … Oecleus fulvidorsum Ball
11′- Middle and right process clearly unequal in stoutness, middle process slender and right process stout in ventral view (fig. 31) .. Oecleus balli Kramer

Figs. 28-30, Oecleus fulvidorsum Ball, from lectotype. Figs. 31-33, Oecleus balli, from type.
12- Aedeagus in lateral view with large round bulge on ventral margin of shaft (fig. 35) … Oecleus campestris Ball
12′- Aedeagus in lateral view with at most a slight convexity on ventral margin of shaft … 13
Kramer 1977 Figs.34-36 O. campestris, 37-39 O. sagittanus, 40-42 O. centronus.
13- Shaft in lateral view roundly expanded on dorsal margin in distal half and anal flap subtriangular in outline (fig. 53) … 14
13′- Without above combination of characters … 15

Kramer 1977 Figs. 43-45 O. subreflexus (California), 46-48 O. subreflexus (Arizonia), 49-51 O. subreflexus (California, allotype of cabazonus), 52-54 O. arnellus.
14- Right process about twice the length of the left process (fig. 52) … Oecleus arnellus B&K
14′- Right process and left process subequal in length (figs. 43, 49) … Oecleus subreflexus Van Duzee
15- Flagellum with two processes … 16
15′- Flagellum with one process … 30
16- Proximal flagellar process in lateral view strongly recurved near the apex (figs. 38, 41). 17
16′- Proximal flagellar process in lateral view straight or only slightly recurved near the apex (figs. 71, 89) … 18
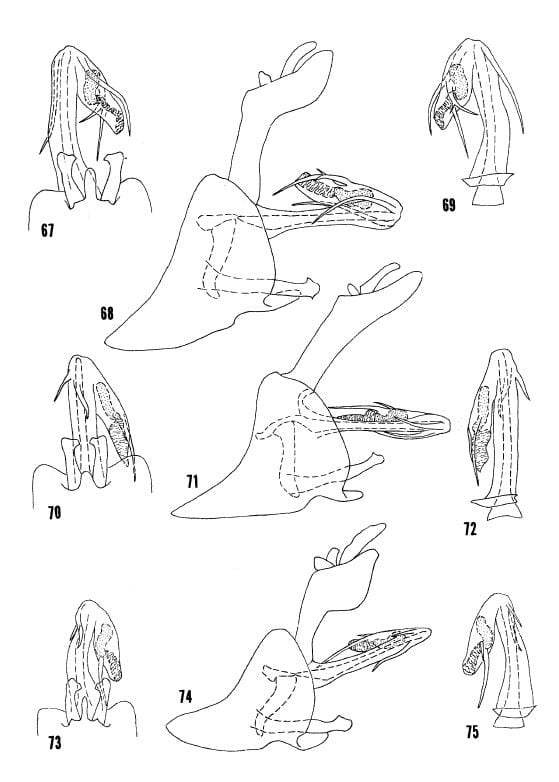
Kramer 1977 Figs. 76-78 O. lineatus, 79-81 O. natatorius, 82-84 O. acutus.
17- Right process short, about one-fourth length of shaft (fig. 37) … Oecleus sagittanus B&K
17′- Right process long, about three-fourths length of shaft (fig. 40) … Oecleus centronus B&K
18- Right process strongly bowed or curved and protruding beyond the outer margin of flagellum at least in part in ventral view … 19
18′- Right process straight or slightly bowed but not protruding beyond the outer margin of the flagellum in ventral view … 22
19- Bases of flagellar processes widely separated (figs. 57, 60), anal flap in lateral view not convex on ventral margin (figs. 56, 59) … Oecleus nolinus B&K
19′- Bases of flagellar processes contiguous, anal flap in lateral view convex on ventral margin … 20

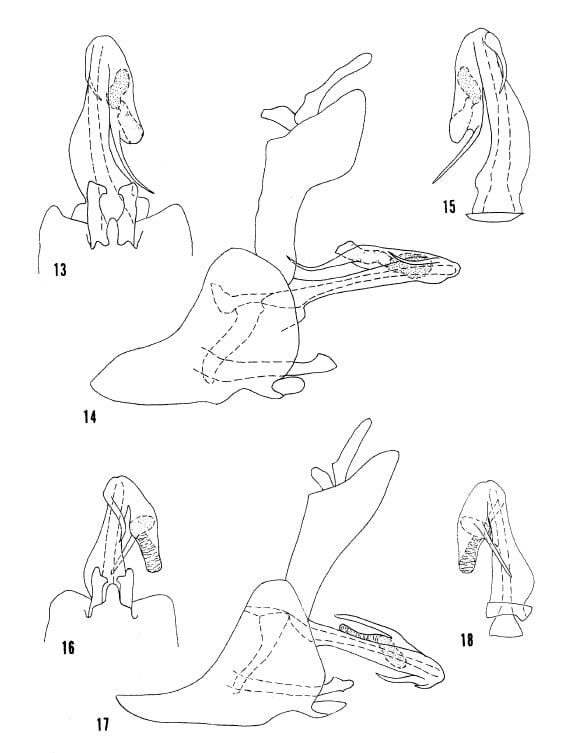
Oecleus nolinus from Tuscon (55-57) and Dona Ana County, New Mexico (58-60)
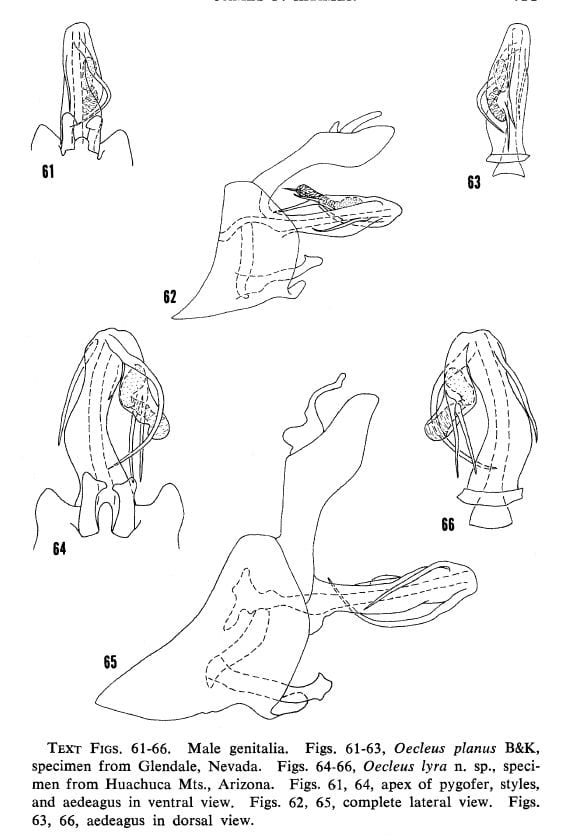
20- Median lobe of pygofer roundly produced at middle, right process abruptly bowed in distal half (fig. 61) … Oecleus planus B&K
20′- Median lobe of pygofer spatulately produced at middle, right process not abruptly bowed in distal half … 21
21- Right process curved to transect flagellar processes in ventral view (fig. 64) … Oecleus lyra Kramer
21′- Right process not curved to transect flagellar processes in ventral view (fig. 67) … Oecleus troxanon Kramer
Kramer 1977 Figs. 67-69 O. troxanon, 70-72 O. perpictus, 73-75 O. chrisjohni.
22- Both right and left processes are half or less the length of the shaft … 23
22′- Either right or left process or both more than half the length of shaft … 25
23- Anal flap elongate and slender (fig. 71), forewing with extensive color pattern (fig. 1) … Oecleus perpicius Van Duzee
23′- Anal flap much broadened distally, forewing without color pattern … 24
24- Median lobe of pygofer narrowly spatulate (fig. 73), flagellar processes of unequal size (fig. 75) … Oecleus chrisjohni Kramer
24′- Median lobe of pygofer roundly spatulate (fig. 76), flagellar processes of equal size (fig. 78) … Oecleus lineatus Ball
Kramer 1977 Figs. 76-78 O. lineatus, 79-81 O. natatorius, 82-84 O. acutus.
25- Right and left processes subequal in length (fig. 79) … Oecleus natatorius Ball
25′- Right process clearly longer than left process … 26
26- Proximal or inner flagellar process minute (fig. 84), median lobe of pygofer broad and roundly produced (fig. 82), (distribution: Haiti) … Oecleus acutus Ball
26′- Proximal or inner flagellar process not minute, median lobe of pygofer spatulately produced, (distribution: Western United states) … 27
27- Anal flap neither strongly convex nor lobed on ventral margin near middle (fig. 89) … Oecleus capitulatus Van D.
27′- Anal flap strongly convex or lobed on ventral margin near middle … 28
Kramer 1977 Figs. 85-87 O. excavatus, 88-90 O. capitulatus, 91-93 O. jenniferae.
28- Both flagellar processes short and barely extending beyond margins of flagellum in dorsal view (fig. 93) … Oecleus jenniferae Kramer
28′- Both flagellar processes long and clearly extending beyond margins of flagellum in dorsal view … 29
29-. Anal flap with irregular lobe on ventral margin distally (fig. 86), right process more than twice length of left process (fig. 85) … Oecleus excavaius Ball
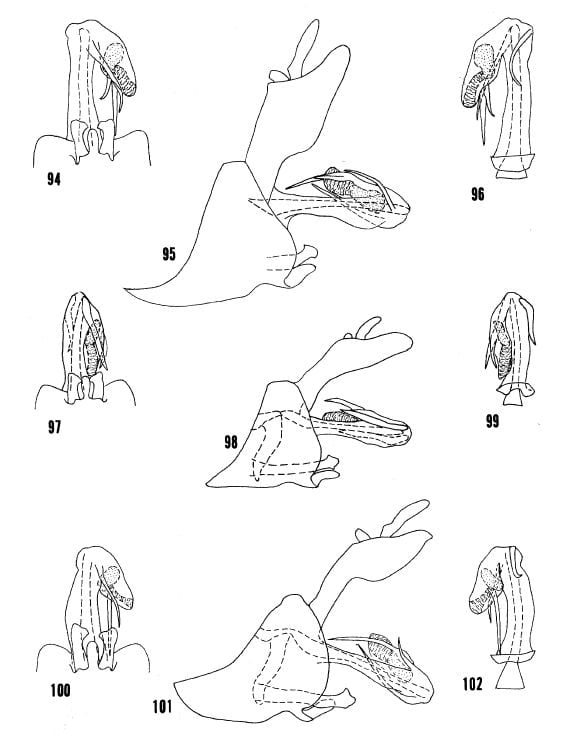
29′- Anal flap broadly convex on ventral margin distally (fig. 95), right process less than twice length of left process (fig. 96) … Oecleus palion Kramer

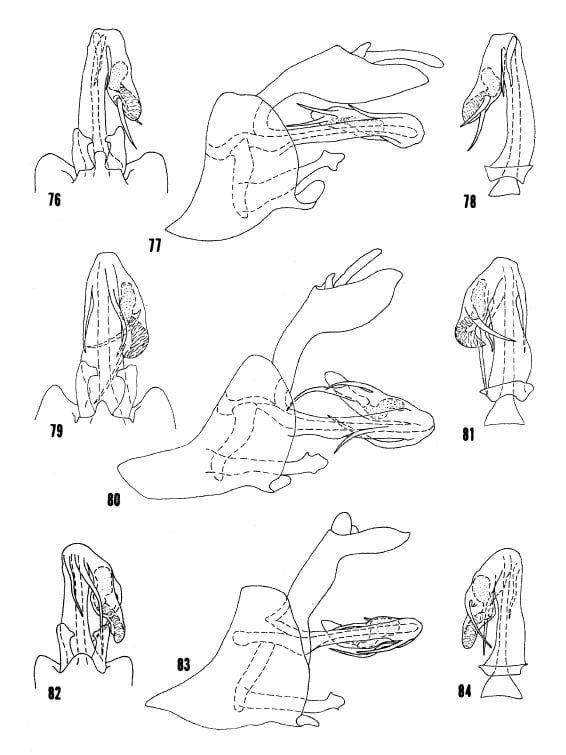
TEXT FIGS (From Kramer 1977). 7-12. – Male genitalia. Figs. 7-9, Oecleus rhion Kramer, from type. Figs. 10-12, Oecleus piperatus B&K, specimen from Sacaton, Arizona. Figs. 7, 10, apex of pygofer, styles, and aedeagus in ventral view. Figs. 8, II, complete lateral View. Figs. 9, 12, aedeagus in dorsal view.

Figs. 94-96, Oecleus palton from type. Figs. 97-99, Oecleus glochin, from type. Figs. 100-102, Oecleus venosus Van Duzee, specimen from Santa Cruz Mts., California.
30- Central portion of median lobe of pygofer in ventral view narrowly spatulate with length at least twice basal width (figs. 97, 100) … 31
30′- Central portion of median lobe of pygofer in ventral view subtriangular or broadly spatulate with length and basal width subequal (figs. 116, 119) … 32
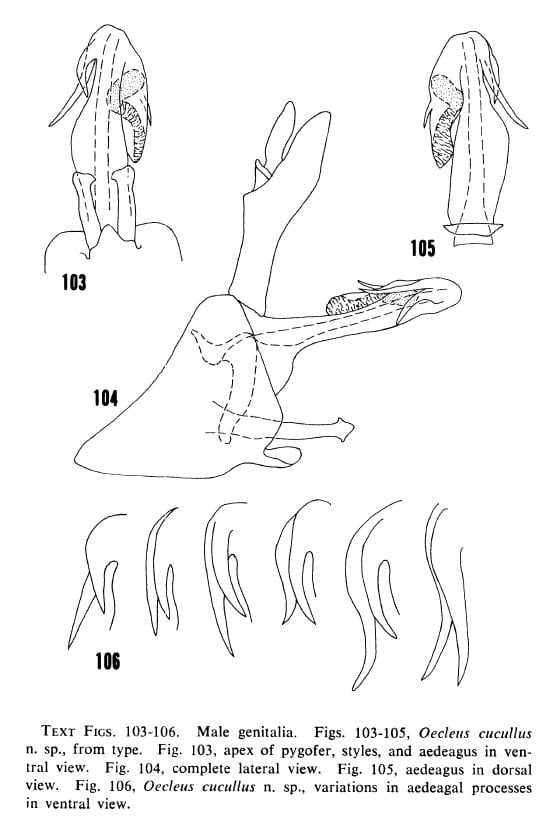
Text Figs. 103-106. Male genitalia. Figs. 103-105, Oecleus cucullus Kramer, from type. Fig. 103, apex of pygofer, styles, and aedeagus in ventral view. Fig. 104, complete lateral view. Fig. 105, aedeagus in dorsal view. Fig. 106, Oecleus cucullus Kramer, variations in aedeagal processes in ventral view.
31- Right process moderately broad with apex acute in ventral view (fig. 97), ventral margin of anal flap not strongly convex distally in lateral view (fig. 98) … Oecleus glochin Kramer
31′- Right process slender with apex needlelike in ventral view (fig. 100), ventral margin of anal flap strongly convex distally in lateral view (fig. 101) … Oecleus venosus Van Duzee
32- Both left and right processes positioned on left margin of shaft in dorsal or ventral view … 33
32′- Only left process positioned on left margin of shaft in dorsal or ventral view … 40
33- Processes crossed at or before their midlengths in dorsal or ventral view … 34
33′- Processes not crossed or subapically crossed in dorsal or ventral view … 36
34- Processes unequal in length and width (figs. 103, 104) … Oecleus cucullus Kramer (in part)
34′- Processes equal or subequal in length and width (figs. 107, 113) … 35
35- Males 5.5-6 mm. long (distribution: Arizona) … Oecleus netron Kramer
35′- Males 4.8-5.1 mm. long, (distribution: California) … Oecleus netrion Kramer (in part)
36- Shaft in dorsal or ventral view with constriction on right side near middle (figs. 113, 115) … Oecleus netrion Kramer (in part)
36′- Shaft not so constricted … 37

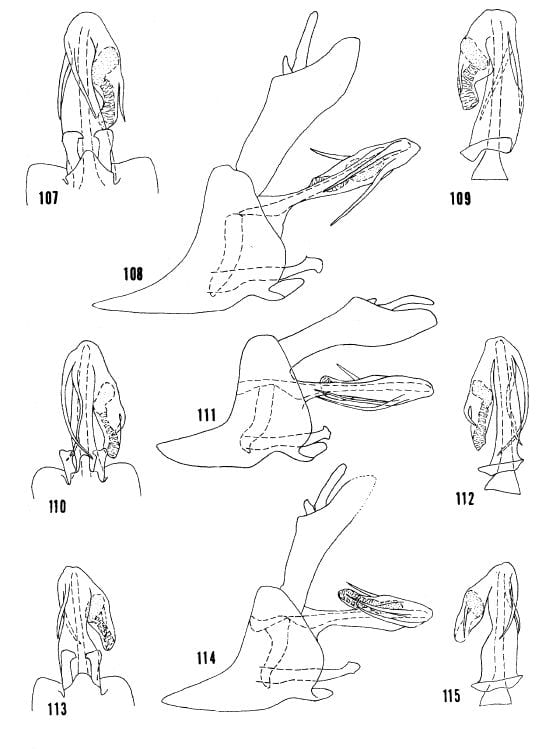
Figs. 107-109, Oecleus netron from type. Figs. 110-112, Oecleus quadrilineatus Van Duzee, specimen from Santa Rita Mts., Arizona. Figs. 113-115, Oecleus netrion from type. Figs. 107, 110, 113, apex of pygofer, styles, and aedeagus in ventral view. Figs. 108, 111, 114, complete lateral view. Figs. 109, 112, 115, aedeagus in dorsal view.
Kramer 1977 Figs. 107-109, O. netron, 110-112, O. quadrilineatus, 113-115 O. netrion.Kramer 1977 Figs. 107-109, O. netron, 110-112, O. quadrilineatus, 113-115 O. netrion.
37- Both processes moderately stout, right process in ventral view in part superimposed on left process for at least half their common length (fig. 106) … Oecleus cucullus Kramer (in part)
37′- Both processes slender, right process in ventral view not superimposed on left process or only slightly so at their extreme bases (figs. 110, 119) … 38
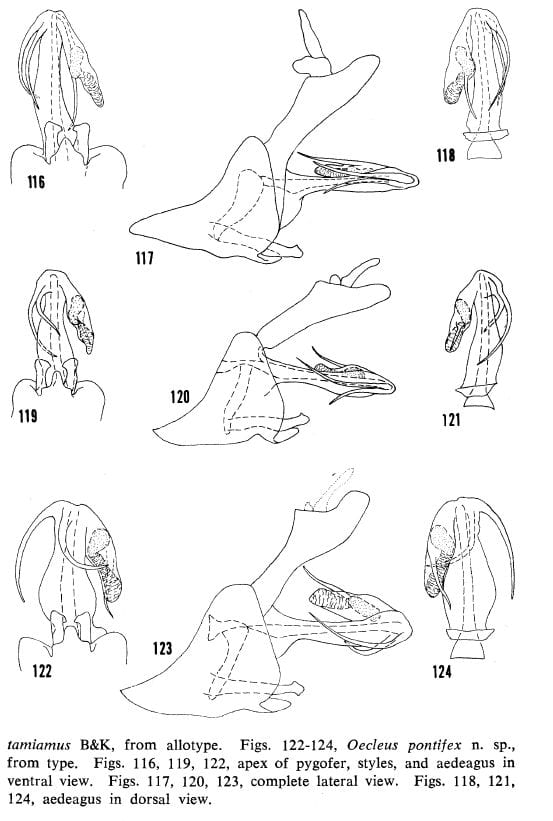
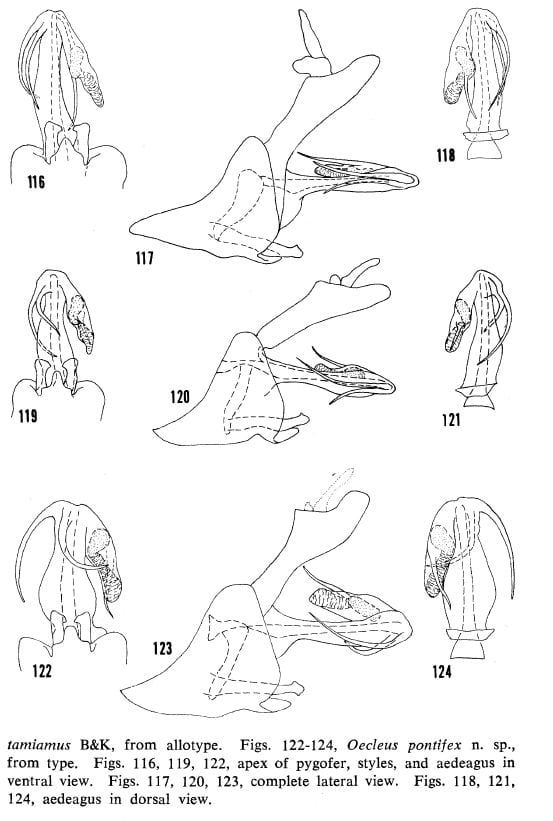
Figs. 116-118, Oecleus borealis Van Duzee, specimen from Sanford, Florida. Figs. 119-121, Oecleus tamiamus B&K, from allotype. Figs. 122-124, Oecleus pontifex from type.
38- Both processes not bowed to the right, right process somewhat undulated, processes usually crossed subapically in ventral view (fig. 110); central and lateral carinae of mesonotal disc black (distribution: Arizona) … Oecleus quadrilineatus Van Duzee
38′- Both processes bowed to the right with similar curvature, processes crossed subapically or not in ventral view (figs. 116, 119); central and lateral carinae of mesonotal disc tawny (distribution: mainly east of Arizona) … 39
39- Head in dorsal view with vertex produced anteriorly for less than greatest width of vertex (distribution: widespread) … Oecleus borealis Van Duzee
39′- Head in dorsal view with vertex produced anteriorly for more than greatest width of vertex (distribution: southcentral Florida) … Oecleus tamiamus B&K
40- Flagellar process long and extending beyond apex of the flagellum in lateral view … 41
40′- Flagellar process short or long but not extending beyond apex of flagellum in lateral view … 42
41- Left process long, strongly projecting beyond left lateral margin of shaft and bowed to the right, right process more narrowly bowed to the right in ventral view (fig. 122) … Oecleus pontifex Kramer
41′- Left process short, not projecting as above and slightly bowed to the left, right process nearly straight or broadly bowed to the left in ventral view (fig. 125) … Oecleus augur Kramer
Kramer 1977 Figs. 125-127, O. augur, 128-130, O. epetrion, 131-133 O. patulus.
42- Left process slender, needlelike and obliquely projecting away from left margin of shaft in ventral view (fig. 128) … Oecleus epetrion Kramer
42′- Left process stouter, not needlelike, not projecting as above in ventral view . 43
43- Right process in ventral view unusually broad basally and broadly curved to the left (fig. 131) … Oecleus patulus Kramer
43′- Right process in ventral view not as above … 44
44- Left process in ventral view with apex not reaching midpoint of bulge on left side of shaft (figs. 134, 137, 143, 146), or if so, right process obliquely directed to right (fig. 140) … Oecleus productus Metcalf
44′- Left process in ventral view with apex reaching to or beyond midpoint of bulge on left side of shaft, right process straight or curved to left (figs. 149, 152, 155, 158) … Oecleus obtusus Ball

Kramer 1977 Figs. 134-136, O. productus (Arizona), 137-139, O. productus (Georgia), 140-142 O. productus (Utah).
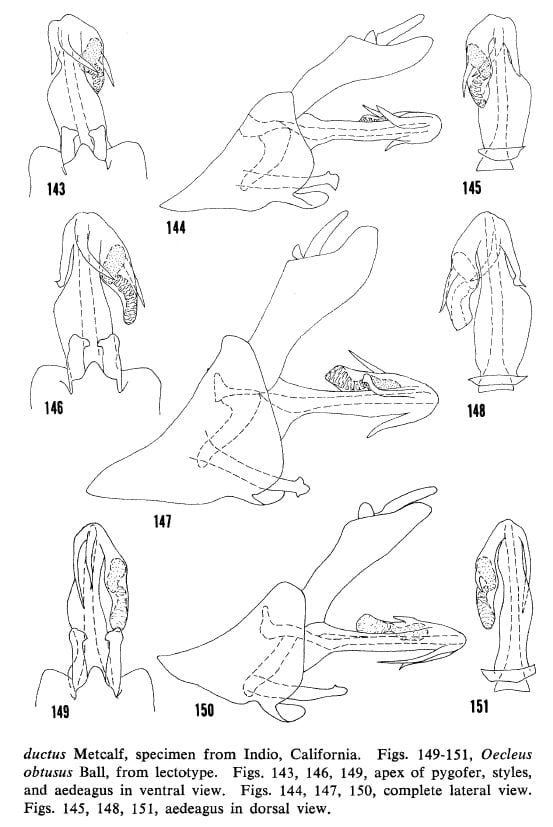
Kramer 1977 Figs. 143-145, O. productus (Arizona), 146-148, O. productus (California), 149-151 O. obtusus.
Kramer 1977 Figs. 152-154, O. obtusus (California), 155-157, O. obtusus (Nevada), 158-160 O. obtusus (Arizona).

Oecleus from Caldwell, 1944. 1 O. pellucens; 2 O. dibius; 3 O. parallelus; 4 O. apterapunctatus; 5 O. concinnus; 6 O. minimus; 7 O. infuscatus; 8 O. delongi; 9 O. constrictus; 10 O. cephalicus, 11 O. spatulatus, 12 O quinquilineatus, 13 O. decens.

Oecleus sp (Louisiana) (may be borealis)(all photographs by Kimberley Shropshire, University of Delaware)

Oecleus snowi (Salvador Vitanza, Extension Entomology in El Paso County)
Online resources
3i.
EOL.
Bugguide.
Discover Life.
FLOW.
Hoppers of North Carolina.
Kunzweb Gallery.
American Insects.
BOLD.
Collecting
Sweeping hosts, or at lights.
Molecular resources
Genbank has data for Oecleus productus, O. perpictus and several Oecleus sp.; Barcode of Life has data for 5 species (Oecleus borealis, O. metatorius, O. nolinus)
Selected references
Ball, E. D. 1902c. Some new North American Fulgoridae. Canadian Entomologist 34: 147-157.
Ball, E. D. 1905a. Some new Homoptera from the south and southwest. Proceedings of the Biological Society of Washington 18: 117-120.
Ball, E. D. 1937c. Some New Fulgoridae from the Western United States. Bulletin of the Brooklyn Entomological Society 32(5):171-183.
Ball, E. D. and P. Klingenberg. 1935. The genus Oecleus in the United States (Homoptera: Fulgoridae). Annals of the Entomological Society of America 28:193-213.
Barrantes, E.A.B., M.A.Z., Echavarria, C.R. Bartlett, E.E. Helmick & B.W. Bahder. 2022. A new species of Oecleus (Hemiptera: Auchenorrhyncha: Fulgoroidea: Cixiidae) from the Caribbean coast of Costa Rica and a reassessment of the generic status of Nymphomyndus. Zootaxa 5133(4): 509–526. https://doi.org/0.11646/zootaxa.5133.4.3
Bartlett, C. R., L. B. O’Brien and S. W. Wilson. 2014. A review of the planthoppers (Hemiptera: Fulgoroidea) of the United States. Memoirs of the American Entomological Society 50: 1-287.
Bartlett C. R., E. M. Dos Passos, F. Gonçalves da Silva, L. E. C. Diniz and M. Dollet. 2018. A new species of Oecleus Stål (Hemiptera: Fulgoroidea: Cixiidae) from coconut in Brazil. Zootaxa 4472(2): 358-364.
Caldwell, J. S. 1944. Notes on Oecleus Stål (Homoptera: Cixiidae). Entomological News 8: 174-176, 198-202.
Emeljanov, A. F. 2007. New and little known taxa of the family Cixiidae (Homoptera, Fulgoroidea). Entomologicheskoe Obozrenie 86(1): 107-131.
Emeljanov, A. F. 1989. To the problem of division of the family Cixiidae (Homoptera, Cicadina). Entomologicheskoe Obozrenie 68(1): 93-106. [Russian] [Translated to English. 1989. Entomological Review 68(4): 54-67]
Foster, D.E. , D.N. Ueckert and C.J. Deloach. 1981. Insects associated with broom snakeweed [Xanthocephalum sarothrae] and threadleaf snakeweed [Xanthocephalum microcephala] in west Texas and eastern New Mexico. Journal of Range Management 34(6): 446-454.
Fowler, H. G. and W. G. Whitford. 1985. Host plant ovipositional preferences and usage by Oecleus nolinus (Homoptera: Cixiidae). Southwestern Naturalist 30(4): 610-611.
Fowler, W. W. 1904. Order Rhynchota. Suborder Hemiptera-Homoptera. (Continued). Biologia Centrali-Americana; contributions to the knowledge of the fauna and flora of Mexico and Central America 1: 77-84, 85-124.
Fowler, H. G. and W. G. Whitford. 1985. Host plant ovipositional preferences and usage by Oecleus nolinus (Homoptera: Cixiidae). Southwestern Naturalist 30(4): 610-611.
Kramer, J. P. 1977. Taxonomic study of the planthopper genus Oecleus in the United States (Homoptera: Fulgoroidea: Cixiidae). Transactions of the American Entomological Society 103: 379-449.
Habeck, D. 1988. Insect detection, Oecleus borealis van Duzee. Tri-Ology 27(11): 6.
Metcalf, Z. P. 1936a. Fascicle IV. Fulgoroidea. Part 2. Cixiidae. General Catalogue of the Hemiptera (Smith College, Northhampton, Massachusetts) 4(2): 1-269.
Muir, F. A. G. 1922. New Malayan Cixiidae (Homoptera). Philippine Journal of Science 20(1): 111-121.
Myrie, W., E. E. Helmick, C. R. Bartlett, A. Bertaccini and B.W. Bahder. 2019. A new species of planthopper belonging to the genus Oecleus Stål, 1862 (Hemiptera: Fulgoroidea: Cixiidae) from coconut palm (Cocos nucifera L) in Jamaica. Zootaxa 4712(1): 127–137. https://doi.org/10.11646/zootaxa.4712.1.9
Myrie, W., B.W. Bahder, E.E. Helmick, & C.R. Bartlett. 2022. Transmission trials of lethal yellowing phytoplasmas carried out with Oecleus mackaspringii. Phytopathogenic Mollicutes 12 (1): 76. [abstract] https://doi.org/10.5958/2249-4677.2022.00041.X
Palmer, W. A. and K. R. Pullen. 1995. The phytophagous arthropods associated with Lantana camara, L. hirsuta, L. urticifolia, and L. urticoides (Verbenaceae) in North America. Biological Control 5: 54-72.
O’Brien, L. B. 1982. Two new species of Oecleus from Texas and Arizona (Homoptera: Fulgoroidea: Cixiidae). Southwestern Entomologist 7(4): 252-254.
Spinola, M. 1839a. Essai sur les Fulgorelles, sous-tribu de la tribu des Cicadaires, ordre des Rhyngotes. Annales de la Société Entomologique de France 8: 133-337.
Stål, C. 1862a. Novae vel minus cognitae Homopterorum formae et species. Berliner Entomologische Zeitschrift 6: 303-315.
Thompson, C. R. 1976. Associations of ants with Cixiidae (Homoptera): I. Nylanderia melanderi and Oecleus borealis II. Aphaenogaster carolinensis and Oliarus vicarius. University of Florida, Gainesville, Florida. Thesis.
Thompson, C. R. 1984. Association of Paratrechina arenivaga (Hymenoptera: Formicidae), with nymphs of Oecleus borealis (Homoptera: Cixiidae). Journal of the New York Entomological Society 92(1): 35-41.
Wilson, S. W. and J. E. McPherson. 1979. The first report of the occurrence of Oecleus chrisjohni and O. epetrion in Illinois (Homoptera: Fulgoridae: Cixiidae) Great Lakes Entomologist 12(4): 227-228.
Wilson, S.W., J. H. Tsai and C. R. Thompson. 1983. Descriptions of the Nymphal instars of Oecleus borealis (Homoptera: Fulgoroidea: Cixiidae). Journal of the New York Entomological Society 91(4):418-423.
Van Duzee, E. P. 1912a. Hemipterological gleanings. Bulletin of the Buffalo Society of Natural Science 10: 477-512.
Van Duzee, E. P. 1923a. Expedition of the California Academy of Sciences to the Gulf of California in 1921 – The Hemiptera (True Bugs, etc.). Proceedings of the California Academy of Sciences (Ser. 4) 12: 123-200.
Van Duzee, E. P. 1925b. New Hemiptera from Western North America. Proceedings of the California Academy of Sciences 14: 391-425.
Van Duzee, E. P. 1929d. A new Oecleus (Fulgoridae). Pan-Pacific Entomologist 5: 173. [Oecleus perpictus, Cixiidae]
Wilson, S. W., J. H. Tsai and C. R. Thompson. 1983. Descriptions of the Nymphal instars of Oecleus borealis (Homoptera: Fulgoroidea: Cixiidae). Journal of the New York Entomological Society 91(4):418-423.
Wilson, S. W., C. Mitter, R. F. Denno and M. R. Wilson. 1994. Evolutionary patterns of host plant use by delphacid planthoppers and their relatives. In: R. F. Denno and T. J. Perfect, (eds.). Planthoppers: Their Ecology and Management. Chapman and Hall, New York. Pp. 7-45 & Appendix.