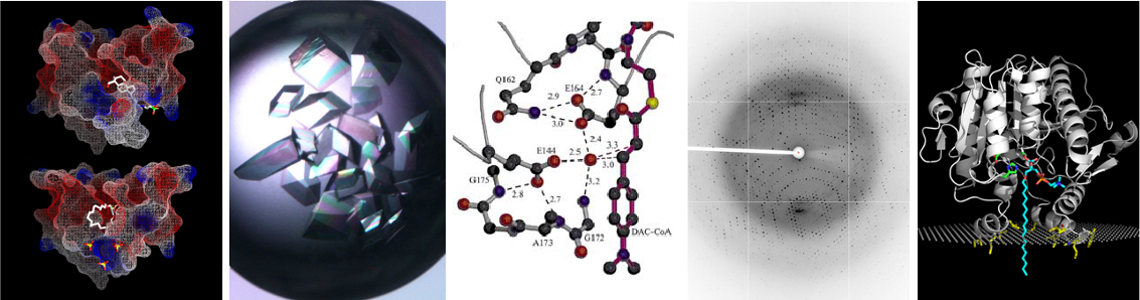
Structural Enzymology:
Current Research
We are exploring the fascinating catalytic power of enzymes with a focus on two related areas of interest:
(i) X-ray crystallography is used to solve structures of substrate complexes and intermediates of enzyme catalyzed reactions and
(ii) understanding the role of evolved ordered enzyme motions in catalysis. This work will provide more thorough answers to the questions: how do precise interactions of all the active site side chains, metal ions, bound water molecules and intermediates contribute to catalysis? What does the transition state look like for a chemical step between two observable enzyme-intermediate structures? How do evolved ordered motions of the entire enzyme play a part in catalysis?
 Structural Enzymology: We are pursuing the structural determination of the mechanisms of several classes of enzymes and with a range of project goals. This work entails solving de novo structures of medically significant enzymes and taking the new structures a step further in a quest to elucidate each enzyme’s mechanism. In one project, we are focusing on a group of human HDL and LDL associated enzymes that have direct links to atherosclerosis. These peripheral membrane proteins are of medical interest due to their connections with signal transduction and lipid remodeling, related to heart disease. Additionally, one of the systems currently under study, called PAF acetylhydrolase (PAF-AH), is also a promising bioscavenger for the detoxification of organophosphate neurotoxins.
Structural Enzymology: We are pursuing the structural determination of the mechanisms of several classes of enzymes and with a range of project goals. This work entails solving de novo structures of medically significant enzymes and taking the new structures a step further in a quest to elucidate each enzyme’s mechanism. In one project, we are focusing on a group of human HDL and LDL associated enzymes that have direct links to atherosclerosis. These peripheral membrane proteins are of medical interest due to their connections with signal transduction and lipid remodeling, related to heart disease. Additionally, one of the systems currently under study, called PAF acetylhydrolase (PAF-AH), is also a promising bioscavenger for the detoxification of organophosphate neurotoxins.

Recently, we have solved the crystal structure of human senescence marker protein 30 (SMP30), and are exploring this enzyme. We use a combination of protein expression, site-directed mutagenesis, kinetics, homology modeling and X-ray crystallography to understand the relationship between structure and function, with a goal of either inhibiting detrimental activities or designing more specific beneficial catalytic activities.

Another related area of interest is to explore how enzymes may have evolved directed motions that are correlated to the reaction coordinate and are essential for their full catalytic power to be achieved. We are attempting to support this hypothesis using dynamic information from anisotropic temperature factor analysis of high resolution X-ray diffraction data. Currently, we are working with a thermophilic protease called thermolysin. This work is an essential component of a complete understanding of enzyme mechanisms, how to create novel catalysts and how to develop selective and potent inhibitors of enzymes.